Activists Are Urging the FDA to Break the Patent on Truvada. Here's Why
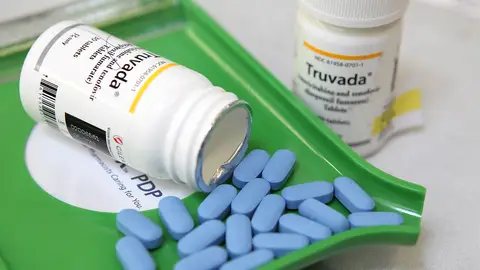
Today, activists nationwide are demanding that the government and drug manufacturers break the patent on the life-saving HIV prevention drug pre-exposure prophylaxis (PrEP), also know by its brand name, Truvada.
According to the CDC, regular PrEP use can reduce the risk of contracting HIV by up to 92%. It's been billed as a "miracle drug" by HIV/AIDS activists and even given out for free in certain states and municipalities with high rates of new HIV infections. But the global manufacturer of Truvada, Gilead Sciences, hiked up the price of the drug by a staggering 25,000%, charging patients without insurance around $1,600 for a 30-day supply of a drug that costs $6 to make per month.
Though the FDA approved a generic version of Truvada last June, Gilead Sciences' drug patents have stalled any forward progress in making PrEP more accessible. The generic is available overseas, but not in America.
Now, HIV/AIDS activists from BreakThePatent.org have declared today, July 17, the first-annual HIV Prevention Day. Advocacy groups like ACT UP New York and the Public Citizen's Access to Medicines are staging digital and in-person demonstrations to urge the FDA and Gilead Sciences to break the patent on Truvada.
"If there is any example of the dysfunction in the American pharmaceutical system, it is this case," ACT UP member James Krellenstein told The Chicago Tribune. "We have the most effective tool for ending the HIV epidemic, and one reason we're unable to scale up is because it costs so [much] unnecessarily."
There's even an online petition circulating with the demand that the National Institutes of Health end the patent on Truvada.
In an age where the president of the United States doesn't even know the difference between HIV and HPV (and fired the nation's entire HIV/AIDS council), HIV awareness and prevention is more vital than ever. Visit BreakThePatent.org to sign the petition and learn more.





